Fish in the Bay – September 2020, Part 2: Shimofuri Special Report and Benthic Bugs.
This post is a cleanup of a few ongoing issues.
- The Shimofuri Goby problem. As discussed many times, Shimofuri Gobies and Chameleon Gobies are hard to distinguish in the field. We know we have both in Lower South Bay. This study is an attempt to discern the relative abundances.
- Shimofuri/Chameleon Goby color change experiments. These two gobies are reported to change colors at will. This must be studied!
- Bad Molluscs* and other interesting critters from the muddy or rocky bottom.
*Mollusc can be spelled either Mollusc or Mollusk. I prefer the international spelling.
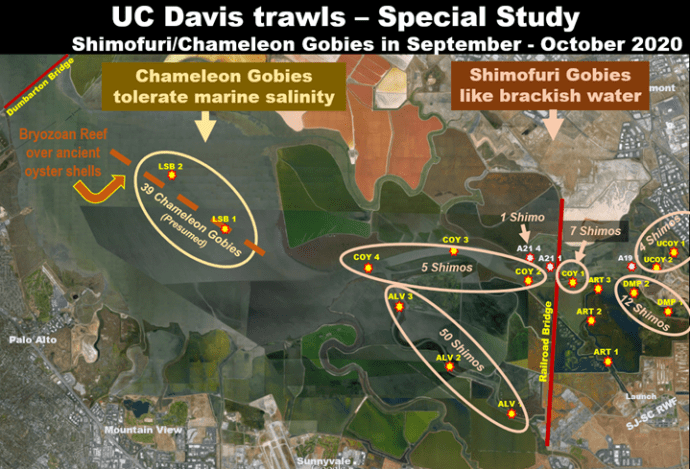
Map showing combined September and October catches of Chameleon and Shimofuri Gobies. – Approximate alignment of the bryozoan & oyster shell hard substrate bottom is also indicated.
1. Gobies in general.
Intro: Gobies are small bottom-dwelling fishes. Males occupy cervices or tunnels or build their own burrows that serve as nests for tending eggs. After spawning, males guard the eggs until hatch.
Most gobid species are marine. Many are tolerant of, or even adapted to, fresh or brackish water. Eggs tend to be laid in downstream burrows since the fry are less tolerant of freshwater. https://en.wikipedia.org/wiki/Gobiiformes
There are at least seven species of gobies in LSB: 3 native and 4 non-native. (Not counting the native Longjaw Mudsucker which technically is also a “gobid.”)

Yellowfin Gobies were first reported in the California Delta in early 1963. They may have arrived around 1960. https://www.cabi.org/isc/datasheet/95431#tosummaryOfInvasiveness (Chameleon Gobies arrived a couple of years later.) Literature says Yellowfins migrate downstream to spawn in winter. Eggs develop at the base of a “Y-shaped” burrow built in the mudflats.

Almost seven years of monthly Goby data from 20 station in Lower South SF Bay.
For most of the last seven years, April was “Baby Fish” month. The optimal temperature for Yellowfin spawning is reported to be 13 degrees C. That means spawning probably happens around December through February, and then small fry show up around April.
For most of those years a Yellowfin population surge roughly corresponded with Baby Fish month. But, Yellowfins did not surge in 2019, and numbers remain lower than average.
Yellowfins probably compete head-to-head with similarly-sized, and also non-native Shokihaze, Chameleon, and Shimofuri Gobies. There has been a small rise in Shimofuri numbers in recent years, but the Shimo rise has been very small in comparison to the yellowfin crash. Unless Shimos are particularly voracious eaters of yellowfin eggs or larvae (which could be possible), I doubt their small numbers make an impact.

Cheekspot and Arrow Gobies are the tiny natives. We discovered a few years ago that not many people take pictures of them. These photos are shown to improve field identification.

Arrow Goby from Alv3, 12 September 2020.
It remains difficult to reliably distinguish Cheekspots from Arrows by naked eye. The distinguishing marks are often too subtle to see without magnification.
The Bay Goby is the other native goby. We saw a few babies earlier this year, but none since then.
2. Adult Chameleon / Shimofuri Goby Identification.

Chameleon Gobies, LSB2, 12 Sept 2020.
The large Chameleon Goby at top was caught in September. He is accompanied by five younger ones. We rarely catch identifiable Chameleon Gobies, so this was a great opportunity to sharpen our fish identification skills.
According to the National Exotic Marine and Estuarine Species Information System (Nemesis) weblink: “While guarding eggs, males become predominantly black (Boltachev and Karpova 2010).” Therefore, our large Chameleon may be a male who was guarding eggs or ready to do so.
- Chameleon Gobies glue their eggs to the walls of caves in hard substrate. They brood multiple times over the warm season.
- The smaller fish in this photo cannot be identified. Chameleon and Shimofuri Gobies are indistinguishable when young. … But, the small fish were LIKELY Chameleon Gobies as well. Salinity was 21.5 ppt. That is the upper salinity threshold for Shimo survival.

The large Chameleon changed color! He lightened up over the few minutes that we watched him.
Chameleon Goby (Tridentiger trigonocephalus) came from East Asia. It was first discovered in California at Los Angeles Harbor in 1960. As the name implies, Chameleon Gobies change color from dark, almost black, to light tan with stripes. https://www.fws.gov/fisheries/ANS/erss/uncertainrisk/ERSS-Tridentiger-bifasciatus-final-October2019.pdf
- Shimo, Chameleon, and Shokihaze gobies are all members of the “Tridentiger genus.” The name means tipletooth gobies because of three cusps they have on their outer teeth. https://en.wikipedia.org/wiki/Tripletooth_goby .
- Shimos and Chameleons are particularly closely related: genetic first cousins, so to speak. This is why they are so hard to distinguish!

Chameleon Goby from July. I now realize that this photo originally shown in the July blog, at the time tentatively identified as Shimofuri, was actually a Chameleon:
- Chameleons have no spots under the jaw.
- Chameleons have a white margin (very faint) on second dorsal and anal fins.
- Chameleon head is boxy or bulbous in comparison to body. (in water – out of water they all buccally respirate!)
Apparently, Chameleon Gobies can range into fairly fresh water. This Chameleon was caught in 12.5 ppt salinity.

Shimofuri Goby (Tridentiger bifasciatus) is also a native of NE Asia. It was first identified in SF Bay in 1985. Shimofuris (Shimos) also change color.
- For many years, Chameleons and Shimofuris were considered the same species. Akihito, the 125th Emperor of Japan was the lead researcher who differentiated these two species in 1989: “Akihito and Sakamoto (1989) resurrected this species after over 50 years of synonymy with T. trigonocephalus.” https://www.fws.gov/fisheries/ANS/erss/uncertainrisk/ERSS-Tridentiger-bifasciatus-final-October2019.pdf
- Shimofuri Gobies do not tolerate marine salinities (Matern and Fleming 1995). https://invasions.si.edu/nemesis/browseDB/SpeciesSummary.jsp?TSN=171912

Shimos at Coy1, 11 Oct 2020. Adult with two young ones.
How to spot a Shimo:
- Shimos have many smaller spots across the cheeks and under the jaw. The word “Shimofuri” means “marbled” in Japanese. This is a reference to the marbled face.
- Shimos have an orange margin on the second dorsal and anal fins. (This is harder to see.)
- Shimo head tapers smoothly to the body. The chameleon head has a boxier or more bulbous appearance in water. (Out of water, both Shimos and Chameleons buccally respirate by puffing their cheeks out, so they both will look bulbous headed.)
3. Baby Chameleon / Shimofuri Goby Identification.

Officially, trying to distinguish Baby Chameleons from Baby Shimos is a lost cause. The human eye cannot see microscopic differences when these fish are much smaller than about two inches.

Baby Chameleons & Shimos resemble adults in the light tan with stripes morph. This is the Chameleon/Shimo default color scheme. I would guess that adults put on dark colors as territorial or courtship displays, or for camouflage.

Baby Chameleon Gobies have fewer spots on the cheeks and NO spots under the jaw.

Baby Chameleons appear to have more angular 2nd dorsal and anal fins. The silhouette of those fins follows a straight, not curved, line. Chameleon anal fins also have a consistent striped pattern: clear, red, clear, black, white margin.

Baby Shimos have many spots on and under the cheeks, like the adults. The 2nd dorsal and anal fins show the orange margins, but this would not be visible to the naked eye. The head is a little more tapered, but again it is too tiny to see in the field.
Generally, naked-eye field identification of tiny baby Chameleons/Shimos will always be hit or miss. Shimos cannot tolerate marine salinity: that remains our best clue when they are too tiny to identify.
4. Shimofuri Goby Color Change Experiment – first two trials.

Shimo Color Change Trial #1: This pair of adult Shimos was caught in Dump Slough, far upstream from the main part of the Bay. Nonetheless, the water was still moderately salty, 15 to 18 ppt, where we caught them.
Again, the orange margins on the rear fins, plus more spots on the face tell us that these are Shimos.

Same two Shimofuris a few minutes later.
In water, Shimofuris have a more smoothly tapered body shape. Chameleons have a boxy or bulbous head. Just remember: if you take them out of water, their heads will swell as they buccally respirate.
Side note: Shimofuris also have a very small iridescent blue patch on the rear edge of their operculum (gill covering). The blue patch is just in front of the base of each pectoral fin.

Same Shimos after 9 minutes.
We watched this pair of Shimos for several minutes to test their color-changing abilities. They seemed to get a little lighter as time passed. (Were they both females??) But, alas, the changing light angle and background in these photos rendered this trial inconclusive.

Shimo Color Change Trial #2: A different pair of Shimos caught in Dump Slough the following month. Right off the bat, we could see that these two fish were avoiding each other. At first, they consistently lined up tail-to-tail in the crowded Photarium.

Romantic Shimos?
The two Shimos were placed in a relatively dark and quiet corner. After roughly 20 minutes, the smaller Shimo had lightened up to tan color. These Shimos were also no longer avoiding each other, but rather hanging kind of close together.
Light versus dark coloration may be a sign of submission/dominance. Light tan with stripes equals baby colors; what could be less dominant than that? Perhaps the smaller Shimo was female? Would Shimos become amorous so quickly in a Photarium?

Angry Shimos?
The Photarium with two Shimos was moved back into bright sunlight. Smaller Shimo immediately began to darken following this disturbance. The photo above shows both adults equally dark 12 minutes later.
The friendly relationship that had developed between these two Shimos was apparently broken once the Photarium was jostled and moved. The simultaneous addition of a third smaller Shimo to the tank probably did not help either.
Conclusion: Adult Shimos can turn dark at will. The color change occurs over less than several minutes. Light tan with dark stripes is the default natal look. The darker look may be the adult aggressive appearance. More investigation needed!
5. Benthic Critter Update.
San Francisco Bay is often called “the most invaded estuary in the world. As far as I know, the tiny California Horn Snail may be the only native mollusc shown in this photo essay!

Bad Molluscs upstream. In the collage above, non-native Corbicula and Corbula clams, to the left, are definitely bad. These little clams will suck nearly all food (phytoplankton, rotifers, copepod neonates, etc.) out of the water column when conditions allow.
- Corbicula clams invade freshwater systems. We occasionally catch a few at the foot of Coyote Creek.
- Corbula clams live in brackish waters a little farther downstream. We consistently catch handfuls of Corbulas where Coyote Creek and Guadalupe River meet the sloshing tides.
Not so bad Molluscs. Macoma Clams and Eastern Mud Snails are also non-native but not so bad – as far as I know.
- Macoma clams eat phytoplankton via filter suspension feeding or surface deposit feeding depending on abundance of food. Macomas we catch may be non-native Macoma petalum, or non-native Macoma balthica (aka Limecola balthica) https://accs.uaa.alaska.edu/wp-content/uploads/macomapetalum.pdf or even hybrids with native “Bent-nosed Macoma” (Macoma nasuta). The Bent-nosed Macoma is still prevalent along the coast. Its shells are found in Native American middens in north San Francisco Bay from long ago. https://en.wikipedia.org/wiki/Macoma_nasuta
- Eastern Mud Snails (Ilyanassa obsoleta) eat excess algae off the mud surface. They came from the Atlantic Coast sometime around 1907.

Bad molluscs downstream (plus a few tunicates):
Atlantic Oyster Drill (Urosalpinx cinerea). This is the main reason Lower South Bay may never support oysters again. Oyster Drills were introduced to SF Bay over a century ago when non-native oysters were imported.
- Oyster Drills are very bad molluscs: https://www.sfchronicle.com/science/article/Scientists-battle-alien-snail-in-SF-Bay-in-effort-13162280.php We catch no live oysters in otter trawls. We catch lots of Oyster Drills. That’s all you need to know.
Philine auriformis. This Snotball is more bad news. This inside-out snail eats native snails and worms. It probably eats oysters as well. https://invasions.si.edu/nemesis/calnemo/SpeciesSummary.jsp?TSN=568082#:~:text=Overview%3A,shallow%20and%20deeper%20coastal%20waters
Tunicates (aka Sea Grapes or Molgula manhattensis) are good news as far as I know. They are relatively innocent Chordate filter feeders: https://en.wikipedia.org/wiki/Tunicate
- Tunicates bloom out of nowhere to suck up excess phytoplankton that this marsh produces. But, unlike Corbula Clams, Tunicates cannot tolerate low salinity, so they rarely intrude into the upstream marsh. Rightly or wrongly, I lump Tunicates and Ctenophores together as beneficial quick-blooming phyto-consumers.

California Horn Snail (Cerithideopsis californica) the last remaining native mollusc?
We caught two tiny California Horn Snails in October. I could not tell if these shells were alive or long dead. We have only seen these once before – about a year and a half ago – reported in the blog at that time. The discovery of a live Horn Snail in restored Pond A19 would be quite interesting. https://en.wikipedia.org/wiki/Cerithideopsis_californica
- Bad news: The Japanese Mud Snail, (Batillaria attramentaria) aka “Asian Horn Snail,” has also invaded California. https://en.wikipedia.org/wiki/Batillaria_attramentaria
- Japanese Mud Snails have invaded north Bay. https://www.kqed.org/quest/40/zen-and-the-art-of-mud-snail-eradication
- Japanese Mud Snails are also common in Elkhorn Slough. http://www.elkhornslough.org/research/grf_jeb.htm
- So far, Horn Snail shells collected here in LSB do not have the distinctive “siphonal notch” found on the invading Japanese variety.

Encrusting Bryozoan forms a coral-like reef in the deep channel of Lower South Bay.
Hard Bottom in the Deep Bay.
Encrusting bryozoan (Cryptosula pallasiana) consolidates ancient oyster shell hash into a ‘coral-like’ reef of hard substrate. http://www.exoticsguide.org/cryptosula_pallasiana
Shells and bryozoan create a very different bottom environment along the deep central channel of Lower South Bay. I presume this Coral-like Bryozoan provides desirable shelter for Cheekspot Gobies, Chameleon Gobies, and Plainfin Midshipmen. Rocky hardness also provides anchorage for red algae, tiny anemones, tunicates, and even native oysters too if we did not have so many oyster drills.

More Encrusting Bryozoan was collected in October.
This hard substrate stands in sharp contrast to vast mudflats across most of the rest of Lower South Bay.

Scale Worm. https://en.wikipedia.org/wiki/Polynoidae This is the mystery polychaete that has been bedeviling me for over a year. The first specimens I found had no scales. I wasn’t sure what they were.
- The scales are deciduous! I literally watched scales fall off this worm of September 2020. It is a curious type of armor. High salinity over most of 2020 might have helped bring this marine worm here. We only see one or two of them each year. They will probably always be a rarity this far south in the Bay.
- Scale Worms are among the most common types of invertebrates off most coasts. http://www.wildsingapore.com/wildfacts/worm/polychaeta/scaleworm.htm

Orange-striped Green Anemones on old oyster shells at LSB1 in October.
Orange-striped Green Anemones (Diadumene lineata). We seem to see blooms of this small non-native anemone every year or so. Like most of the other non-natives, these were likely introduced with oyster shipments a century ago. They must anchor to hard substrate; hence we mainly see them on old oyster shells at LSB1. Salinity below 12 ppt kills them, so they cannot colonize much farther upstream. https://www.exoticsguide.org/diadumene_lineata
6. Red Algae Watch.

Cryptopleura, Red Gracilaria, and Ceramium tend to peak in late summer and fall. The same is true for loose clumps of Ulva, a green algae. These are all good food for fish and snails.
Ceramium Mystery. In July and August, we collected huge masses of Ceramium at LSB and Coy4 stations. But, when we trawled in September; Ceramium was gone! COMPLETLY GONE! Where did it go? Why did it bloom so suddenly and for so short a period? Did something eat it? … We shall keep watching.
On to the October report.
 Previous Post
Previous Post Next Post
Next Post